Blogs

6 Challenges in ADME Drug Development
Explore the current challenges in profiling preclinical human ADME through key publications. Can advances in the human relevance of preclinical models improve estimations for safer and more efficient drug development?
Posters

Defining validation criteria for a primary jejunum and primary hepatocyte dual-organ MPS
Improve in vitro to in vivo data translation for drug safety and efficacy with our fluidically-linked dual-organ MPS (microphysiological system); combining two well-characterized human Gut/Liver MPS – the RepliGut® Jejunum and PhysioMimix® Liver MPS, in an interconnected model suitable for enhanced bioavailability profiling.
Blogs

How in vitro human Gut/Liver models increase confidence in ADME estimations before human trials
This blog explores a world where novel multi-organ in vitro models, tiny replicas of the human intestine and liver, provide the key to unlocking a deeper understanding of how we Absorb, Distribute and Metabolize drugs.
Read more to discover why accurately estimating human ADME remains a challenge and a potential solution that enables greater confidence in lead candidates and a reduced risk of identifying poor oral bioavailability during first in human studies.
Application notes

Connecting the gut and liver
A human-relevant dual-organ microphysiological system connecting the gut and liver for preclinical profiling of oral bioavailability
Efforts to improve the in vitro to in vivo translation of drug efficacy and safety data has led to the emergence of more human relevant microphysiological systems (MPS). Multiple, fluidically linked MPS can be linked to form multi-organ systems that simulate human processes which can be utilized to improve ADME and bioavailability estimations. ADME and bioavailability are central in determining the safety and toxicology profiles of compounds and are therefore crucial preclinical drug development measurements.
Posters

Connecting the human intestine and liver
A primary jejunum and primary hepatocyte multi-organ MPS for more predictive studies of human drug ADME and oral bioavailability
Traditional immortalized intestinal cell lines and suspension hepatocytes have absent or low levels of metabolic enzyme expression, and thus fail to predict first pass human metabolism and oral bioavailability.
Efforts to improve the in vitro to in vivo translation of drug efficacy and safety data has led to the emergence of more human relevant microphysiological systems (MPS) that consist of multiple, fluidically linked organs1.
Articles

Harnessing Microphysiological Systems to Bring Humanized Processes to ADME and Bioavailability Studies
Dr. Abbas discusses the common drawbacks of using conventional methods for ADME studies that can misinform the candidate selection process and dosage figures. He talks about how emerging complementary technologies, such as organ-on-a-chip (OOC), offer human-relevant in vitro preclinical data, and how using OOC can support the determination of human bioavailability to support drug dosing regiments, reduce side effects, and potentially recover flawed therapeutic candidates.
Brochures & Flyers

Organ-on-a-chip Contract Research Services Brochure
Discover our full range of Organ-on-a-chip contract research services including ADME, NASH, DILI and Oncology as well as all the relevant endpoints.
Posters

A primary jejunum and primary hepatocyte multi-organ MPS
Find out how our gut-liver MPS recapitulates the physiological condition enabling oral drug dosing in vitro. This gut-liver model offers a vast improvement in the methods used to study PK or prodrugs.
Articles
ADME Studies: Determining Promising Drug Compounds
Dr Abbas discusses factors that can affect the outcome of an ADME study, signs that a drug compound shows promise, red flags, and key parameters to determine safety and efficacy.
This article is taken from PharmTech, November 2022.
Articles

How to Keep Breathing – The Future of Inhaled Medication Testing
Dr Emily Richardson discusses the current challenges faced to bring inhaled therapeutics to the market and the potential of Organ-on-a-Chip to increase positive outcome by improving ADME drug testing.
This article is taken from International Biopharmaceutical Industry, Summer 2022.
Videos and animations
PhysioMimix Multi-organ System Animation
An introduction to the CN Bio PhysioMimix Multi-organ System. This animation demonstrates how our microphysiological system works, how to create a Gut/Liver-on-a-chip model and an example of its use in determining drug Bioavailability in vitro.
Posters
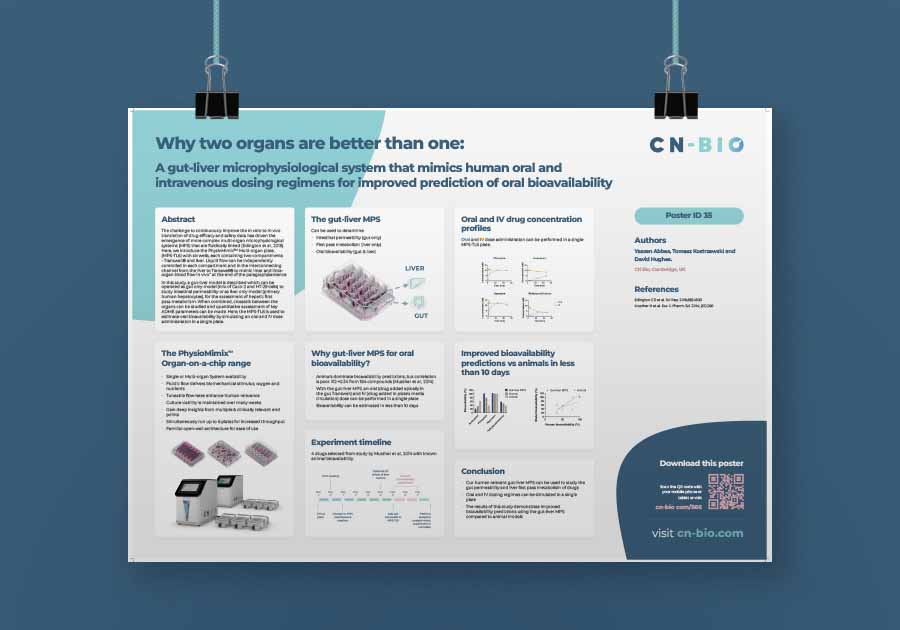
Why two organs are better than one
Abbas, Kostrzewski & Hughes
This poster demonstrates how a gut-liver MPS can improve oral bioavailability predictions by mimicking human oral and IV regimens. Thus improving the in vitro to in vivo translation of drug efficacy and safety.

